This is a guest post by SCM's business developer Fedor Goumans
To further improve the efficiency and life-time of organic light-emitting diodes (OLEDs), ultimately the properties of underlying materials need to be tweaked at the molecular level. In materials science, as in other fields, modeling has become a more wide-spread tool integrated with experiments for a holistic research & development approach.
In particular, high-throughput screening computational screening may considerably reduce experimental costs for synthesizing and testing new materials. At the molecular level there are a few properties that are important for OLED life-time and efficiency. A simplified set up for an OLED device is depicted in Figure 1. The charge mobility and light emission properties can be predicted with computational chemistry, as explained below.
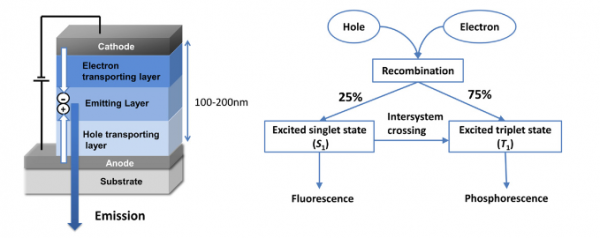
Figure 1: OLED device set-up and fundamental processes. Picture taken from: Y. Suzuri et al., Phosphorescent cyclometalated complexes for efficient blue organic light-emitting diodes, Sci. Technol. Adv. Mater. 15 (2014) 054202
Under working conditions of the OLED, positive charges (holes) from the anode recombine with negative charges (electrons) from the cathode to form excited states: excitons. Excitons can relax to the ground state by emitting light. Relaxation via other pathways typically generate heat and can deteriorate the device.
Electrons and holes have a quantum mechanical property called spin, and the rules of quantum mechanics dictate that, upon recombination, 25% of these excitons are in a so-called singlet state (spins anti-parallel) while 75% forms in the triplet state (spins parallel). In a purely organic material, only the singlet excitons can emit light (fluorescence), again due to the laws of quantum mechanics, thus resulting in a maximum theoretical efficiency of 25%.
In typical present-day devices, transition metal complexes are used which can emit light from the triplet excitons (phosphorescence), and furthermore can harvest the singlet excited states to the triplet state before they emit (intersystem crossing. Both these processes are allowed through the combination of quantum mechanics and relativity, which makes the spin quantum property couple with angular momentum of the electrons orbiting the nuclei. This spin-orbit coupling typically only manifests itself noticeably for the heavier elements (lower rows of the periodic table) where relativity plays a larger role for the faster moving electrons.
Modeling charge mobilities for organic electronics
With computational chemistry the properties of hole and electron mobility can be studied by calculating the propensity (electronic coupling) for the charge to move from one molecule to another (Figure 2).
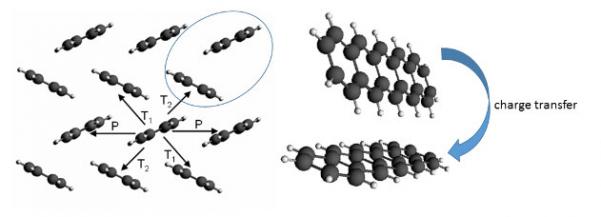
Figure 2: mobility in organic materials can be studied by calculating the hopping probability between different dimers, as shown here for pentacene
With different orientations of neighboring molecules in an organic crystal, charges are more mobile in certain directions than in others. This anisotropicity has been correctly predicted for a set of organic semi-conductors in S.-H. Wen et al., First-Principles Investigation of Anistropic Hole Mobilities in Organic Semiconductors J. Phys. Chem. B 113, 8813-8819 (2009).
Modeling phosphorescence for OLEDs
Phosphorescent lifetimes can be predicted with quantum chemistry codes combining spin-orbit coupling relativistic effects with calculating excited states. Likewise, the rate of intersystem crossing (triplet harvesting) can be estimated.
By modifying the molecule used as a phosphor in OLED devices, a different color can be obtained and in particular the radiative rate can be optimized. Molecular modeling can be used to predict radiative lifetimes of these phosphors, as has been demonstrated by a few companies producing OLED materials (H. Sasabe et al., High-Efficiency Blue and White OLEDs Incorporating a Blue Iridium Carbene Complex, Adv. Mater., 22, 5003-5007 (2010); J. M. Younker and K. D. Dobbs, Correlating Experimental Photophysical Properties of Iridium(III) Complexes to Spin-Orbit Coupled TDDFT Predictions, J. Phys. Chem. C 117, 25714-25723 (2013)).
For optimal OLED performance, a high phosphorescent rate is desirable, and computational chemistry can be used to model this without having to go through laborious experimental procedures first. The computational approach also works for predicting phosphorescent properties of different transition metals, not just the often-used iridium complexes (Figure 3).
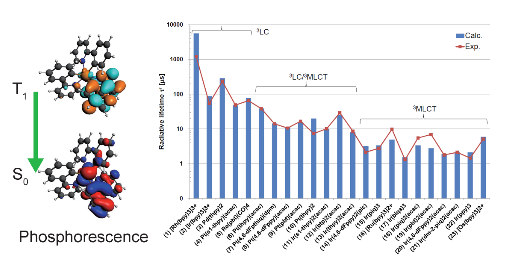
Figure 3: Predicted radiative lifetimes for 23 different transition metal complexes in good agreement with experiment, with the exception of Rh complex Rh(bpy)33+ which also relaxes via alternative pathways (K. Mori et al. Predicting phosphorescent lifetimes and ZFS of organometallic complexes with SOC-TDDFT, Phys. Chem. Phys. Chem.16, 14523 (2014))
Other important molecular processes in OLEDs have to do with non-radiative relaxation of the excitons. In particular the recombination of excitons with charges (exciton-polaron coupling) and other excitons (exciton-exciton annihilation) will reduce luminescent efficiency and can deteriorate the device via heat generation leading to damage at the molecular level. These properties can also be studied with more advanced computational chemistry approaches.
This entry was written by SCM's business developer Fedor Goumans, who has many years of computational chemistry experience in various experimental groups. SCM is a spin-off company from the VU University in Amsterdam, developing the Amsterdam Density Functional (ADF) modeling suite in collaboration with many (academic) partners. ADF has unique capabilities for modeling molecular properties for organic electronics such as described here.

